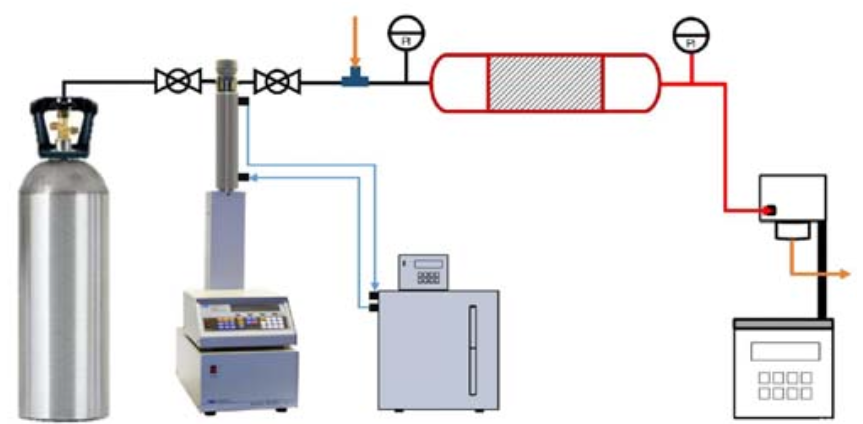
Catalytic CO₂ Conversion and In Situ Spectroscopy at High Pressure
CO₂ is a thermodynamically stable molecule and an increase in reaction pressure is known to be advanta- geous for efficient CO₂ activation as evident from the thermodynamic equilibrium conversion and selectivity calculations (Figure 1). The methanol synthesis pro- cesses are operated typically at about 50–100 bar with low CO₂ conversion efficiency and low methanol selec- tivity, often requiring extensive recycling of unconverted reactants. Besides the thermodynamic and kinetic advantages, high-pressure approaches can facilitate the reduction in reactor volume when reactants and/or products are compressive and handling dangerous chemicals such as hydrogen due to smaller contained volume, giving pos- sible economic as well as safety advantages. To achieve this ambitious goal of performing reactions in contin- uous operation (flow condition) at very high pressure (up to 500 bar), a reactor system using Teledyne Isco 260D syringe pump was developed. Along with the reactor system, the syringe pump was also used in the development and operation of in situ spectroscopic tools to study the phase behavior and reactions under the demanding high pressure operating conditions.